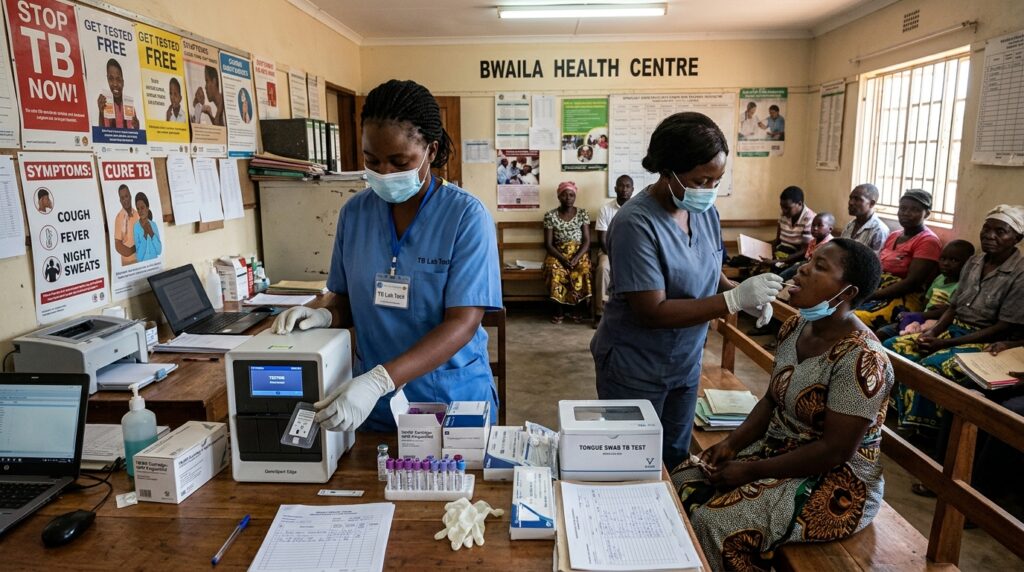
The agency’s March 2026 recommendations on near-point-of-care molecular tests and tongue-swab sampling mark a notable shift toward faster, simpler tuberculosis diagnosis, especially for patients who struggle to produce sputum or reach centralized laboratories.
The World Health Organization has given its backing to a broader set of tuberculosis diagnostic tools in a move that public health specialists say could help bring testing closer to patients, shorten delays to treatment and widen access for groups long missed by conventional approaches.
In recommendations highlighted in March and April 2026, WHO urged countries to expand use of new diagnostics including molecular tests that can be used near the point of care and tongue-swab sampling for certain patients who cannot produce sputum. The changes may sound technical, but they touch one of the most stubborn problems in global health: how to detect tuberculosis quickly enough, and simply enough, to stop people from dying before treatment begins.
That challenge remains enormous. Tuberculosis is preventable and curable, yet it continues to rank among the deadliest infectious diseases in the world. WHO says 1.23 million people died from TB in 2024, including 150,000 people living with HIV, while 10.7 million people fell ill globally. Those figures help explain why even seemingly incremental diagnostic improvements are attracting close attention. In TB control, a better test is not just a laboratory upgrade. It can determine whether a patient is diagnosed in time, whether infection continues to spread within households and communities, and whether already strained health systems can use scarce resources more effectively.
WHO’s latest push is built around a practical premise: TB diagnosis must become easier to deliver where people actually seek care. For years, molecular tests have transformed the field by improving accuracy over smear microscopy, but many systems still depend on centralized platforms, reliable electricity, trained laboratory personnel and the ability to collect sputum. Each of those requirements can become a barrier. A patient may live far from a reference laboratory. A rural clinic may not have the infrastructure for standard molecular workflows. Some patients, including people who are very ill, elderly or living with HIV, may struggle to produce sputum at all.
The newly recommended near-point-of-care nucleic acid amplification tests are intended to help close that gap. According to WHO, these devices can run on battery power, operate in peripheral clinics or mobile services and return results in less than an hour. They are designed to be simpler to use than conventional laboratory-based systems, with fewer infrastructure demands and less need for highly specialized staff. WHO has also said that some of these tools are available at less than half the cost of many existing molecular diagnostics, an important consideration for countries where TB burden is high and funding remains constrained.
That combination of speed, portability and lower complexity is why the recommendation matters. In its March guidance, WHO said near-point-of-care molecular tests on sputum should be used as initial diagnostic tests for TB in adults and adolescents with symptoms or a positive screening result, rather than relying on smear microscopy. It also said that when sputum cannot be obtained, these tests on tongue swabs should be used as initial diagnostic tests for TB. In policy terms, that is significant because it gives programs a clearer route to diagnosing patients who would otherwise be hard to test at all.
Tongue swabs are especially notable because they address one of the oldest frustrations in TB diagnostics: the dependence on sputum. Sputum remains the preferred sample because it generally offers better diagnostic accuracy. But for many patients, producing a sputum sample is difficult, inconsistent or impossible. WHO’s updated guidance recognizes that a simple oral sample can, in selected situations, provide a practical alternative that expands access to testing. The recommendation does not elevate tongue swabs above respiratory samples, and it does not suggest they are equally accurate in all contexts. Instead, it reflects a public health calculation that a somewhat less sensitive but easier-to-obtain sample may be far better than no test at all.
WHO has been careful to frame these changes with nuance. The agency says respiratory samples should still be collected and tested whenever possible because they perform better than tongue swabs. On some low-complexity automated platforms, WHO’s recommendation for tongue-swab use is conditional and based on low-certainty evidence. For the newer near-point-of-care class, however, WHO issued a strong recommendation with moderate-certainty evidence for use of tongue swabs when sputum cannot be obtained. The message is not that old diagnostic logic has been overturned, but that health services now have more flexibility to reach patients who previously sat outside the most effective testing pathways.
That flexibility could be particularly important in decentralized settings. Clinics without advanced laboratory facilities, mobile teams working in remote communities and overstretched urban primary-care centers all face the same broad question: how can TB testing be made quick, feasible and acceptable without losing too much accuracy? WHO’s answer appears to be that no single tool will solve the problem, but a broader diagnostic toolkit can make systems more responsive. In addition to near-point-of-care molecular tests and tongue swabs, the agency has also highlighted sputum pooling as a way to conserve machine time and commodities in severely resource-constrained settings.
For national TB programs, the appeal is clear. Earlier diagnosis can reduce transmission. Faster results can reduce the number of patients lost between testing and treatment initiation. Simpler sample collection can expand reach to populations that currently face repeated barriers. And equipment that works outside central laboratories can shift diagnosis closer to communities where TB burden is highest. These are not abstract advantages. They speak to long-standing weaknesses in the TB cascade of care, where many patients are diagnosed late, diagnosed only after multiple contacts with the health system, or never diagnosed microbiologically at all.
Yet the optimism around the new recommendations comes with important caveats. One is that implementation is always harder than policy. A WHO endorsement does not automatically translate into procurement, training, quality assurance, supply chains, maintenance or reimbursement. Countries will need to decide which platforms fit their systems, how to integrate new sampling strategies into diagnostic algorithms and how to ensure positive patients are linked promptly to treatment and drug-susceptibility testing. WHO itself notes that some near-point-of-care tests do not detect rifampicin resistance, meaning additional testing remains necessary after a positive result.
Another caution is that evidence remains uneven across patient groups. WHO’s recommendation for tongue swabs is focused mainly on adults and adolescents, especially those unable to provide sputum. The agency has been more restrained about broader use in children because evidence is still limited. It has also indicated that for people living with HIV, concurrent testing strategies using other recommended tools may still be preferred where available. In other words, the new options broaden the map, but they do not erase the need for layered, context-specific diagnostic pathways.
Even so, the direction of travel is unmistakable. Global TB policy is moving away from an overly centralized model in which patients must fit the test, and toward one in which the test is more adaptable to the patient and the setting. That may prove one of the most consequential shifts in the field, because TB control has long suffered not only from biological difficulty but from operational friction. Delays, travel costs, weak referral systems and specimen challenges often blunt the impact of technologies that look impressive on paper.
By endorsing tools that can be used in basic peripheral facilities, that can rely on battery-powered devices and that can use tongue swabs when sputum is not available, WHO is signaling that access and practicality are no longer secondary concerns. They are part of diagnostic quality itself.
For all the technical detail, that may be the clearest reason this update deserves attention. Tuberculosis remains a disease of profound inequality, hitting hardest where health systems are weakest and patients have the least room for delay. A diagnostic strategy that reaches people earlier, more locally and with fewer barriers will not end TB on its own. But it could help narrow one of the deadliest gaps in the response: the distance between having the disease and getting a confirmed diagnosis in time.
That is why WHO’s March-April 2026 recommendations stand out as one of the most consequential public health developments of recent weeks. They do not promise a breakthrough in the dramatic sense. What they offer is something more durable and, in global TB control, often more valuable: a practical chance to find more patients sooner, with tools built for the realities of where they live and how they seek care.